|
With minimized electrode passivation, high sulfur utilization (~95%) and stable cycling were achieved using an extremely small surface area cathode (0.9908 m 2 g −1), even without additional Li metal protection. The key advantage of this salt-modification approach is that Li 2S growth chemistry can be modulated without severe deterioration of Li metal electrodes, which is unattainable with high-DN solvent electrolytes. Li 2S growth behaviors were examined critically for three salt anions with different DNs in a conventional 1,3-dioxolane (DOL):1,2-dimethoxyethane (DME) (1:1) solvent. By changing a salt anion into one with a higher-DN, we induced 3D particulate growth of Li 2S. Herein, we suggest a supporting salt anion as an agent to control the electron-donating property of an electrolyte. Thus, practical application of high-DN solvents for Li–S batteries remains challenging 33, 34. For this reason, stable cycling of Li–S cells has not been achieved with high-DN solvent-based electrolytes without advanced Li metal protection. Although high-DN solvents offer advantages, they have the formidable drawback of extreme reactivity with a Li metal electrode. The amended Li 2S morphologies are in line with the electrolyte dependency of lithium peroxide (Li 2O 2) morphology in Li–air batteries electrolytes with high electron donating ability augment the solubility of lithium superoxide (LiO 2) and produce 3D toroidal-shaped Li 2O 2 35. Li 2S films were observed with the low-DN solvent, whereas flower-like Li 2S particles grew with the intermediate and high-DN solvents 34. More recently, variation of Li 2S deposition morphology was reported in response to the solvent DNs. By using a high-DN solvent, dimethylacetamide (DMA), full utilization of sulfur and Li 2S was achieved based on enhanced chemical transformation between Li 2S and soluble PS anions 33. It has been reported that solvents with high Gutmann Donor Number (DN) promoted the redox reaction of sulfur species by stabilizing multiple states of PS anions and altering the kinetic pathway 32. The approaches discussed above feature control of either the cathode material or the structure conversely, modulating the intrinsic chemistry of Li 2S growth by electrolyte optimization could offer a fundamental solution for achieving high sulfur utilization. Nevertheless, the discharge capacity remains limited due to passivation of the conducting network. Macro-pore structures relieve the clogging of ion transport channels by Li 2S deposits. Another option would be employing a three-dimensional (3D) conducting network with macro channels 30, 31. These approaches effectively enhance sulfur utilization and suppress the polysulfide (PS) shuttle however, the electrode passivation issue is not completely resolved during repeated cycles. 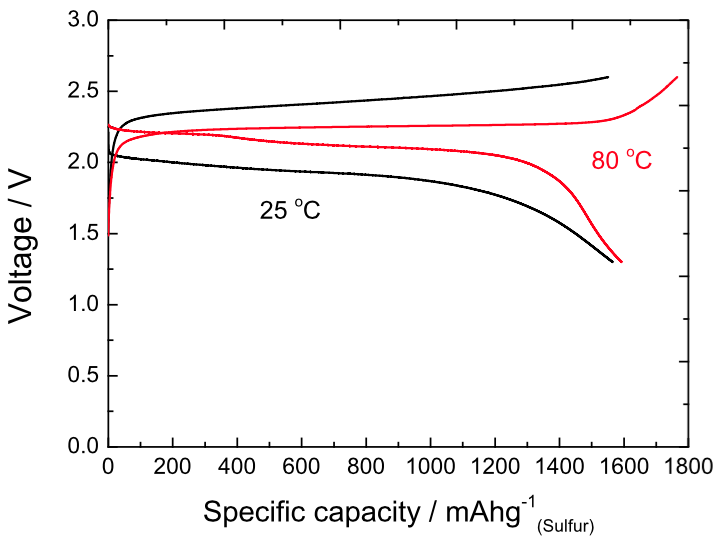
One approach for suppressing electrode passivation is uniform distribution of sulfur species, either by confining them physically in structured host materials such as porous carbons 19, 20, graphene materials 21, 22, and conductive polymers 23, 24 or by chemically adsorbing them onto structures that include transition metal chalcogenides 25, 26, 27 and metal organic frameworks (MOFs) 28, 29. The problem becomes more significant at higher current density and higher areal sulfur loading, hindering rational design of practical Li–S batteries (Supplementary Fig. 
This impedes discharge before complete use of the loaded sulfur 12, 18. Due to its electronic and ionic insulating property, a film of Li 2S covering the cathode causes early electrode passivation and consequent large polarization. In particular, during discharge, uncontrolled electrodeposition of Li 2S, the final discharge product, is a critical obstacle to achieving complete sulfur utilization. These limitations result in low sulfur utilization 12, 13, low coulombic efficiency 14, 15, and fast capacity fading 16, 17 of Li–S batteries. However, practical applications of Li–S batteries are still hampered by intrinsic problems such as the low conductivity of sulfur and lithium sulfide (Li 2S) 3, 4, 5, large volumetric changes of the electrode 6, 7, 8, and dissolution of intermediate lithium polysulfide (LiPS) species during cycling 9, 10, 11. With increasing demand for energy-dense batteries that can overcome limits of conventional lithium (Li) ion technology, lithium–sulfur (Li–S) batteries are considered the most promising candidate because of their high energy density (2600 W h kg −1) and structural similarity with Li-ion batteries 1, 2.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
